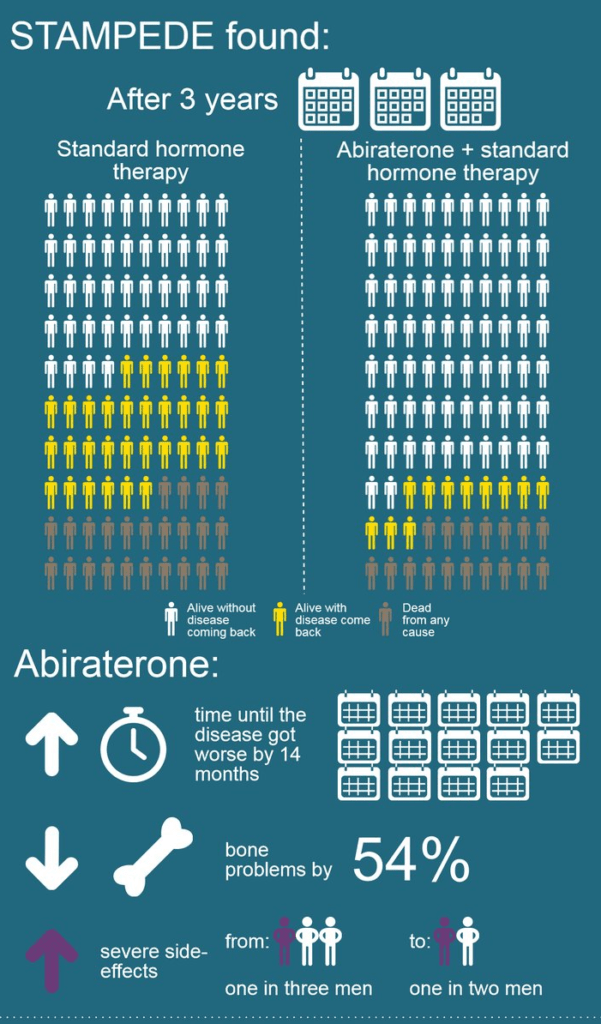
The results of a new study presented at the 2017 ASCO Annual Meeting in Chicago report that abiraterone (Zytiga) improves prostate cancer survival when it is added to hormone therapy at the beginning of treatment for the disease. An improvement of 37 percent was seen in the trial, which was known as STAMPEDE (Systemic Therapy in Advancing or Metastatic Prostate cancer: Evaluation of Drug Efficacy).
These findings are significant because they could make abiraterone a first-line treatment choice along with hormone therapy for men with high-risk locally advanced or metastatic prostate cancer.
Traditionally, abiraterone is used as treatment for advanced metastatic cancer that have not responded to hormone therapy. Abiraterone stops the production of hormones that fuel the growth of prostate cancer.
The STAMPEDE trial involved 1,917 men who were randomly assigned to standard care (hormone therapy for at least 2 years) with or without the addition of abiraterone (1,000 mg plus 5 mg prednisone daily). Men who had locally advanced prostate cancer were allowed to receive radiation therapy. Results were as follows:
- A 70 percent reduction in prostate cancer progression in men who received abiraterone
- An increase in the time (14 months) it took for the disease to become worse
- A 54 percent reduction in bone complications, which is a major problem in men with prostate cancer
- On the downside, there was an increase in severe side effects from 33 percent of men (hormone therapy alone) to 50 percent (hormone therapy plus abiraterone)
According to Professor Nicholas James, chief investigator of the Cancer Research UK-funded study from the University of Birmingham, “These are the most powerful results I’ve seen from a prostate cancer trial…[and] “one of the biggest reductions in death I’ve seen in any clinical trial for adult cancers.”
Read more in our Prostate Cancer Health Center
Reference
Cancer Research UK. Adding abiraterone to standard treatment improves prostate cancer survival by 40 per cent. ScienceDaily 4 June 2017